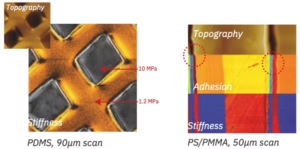
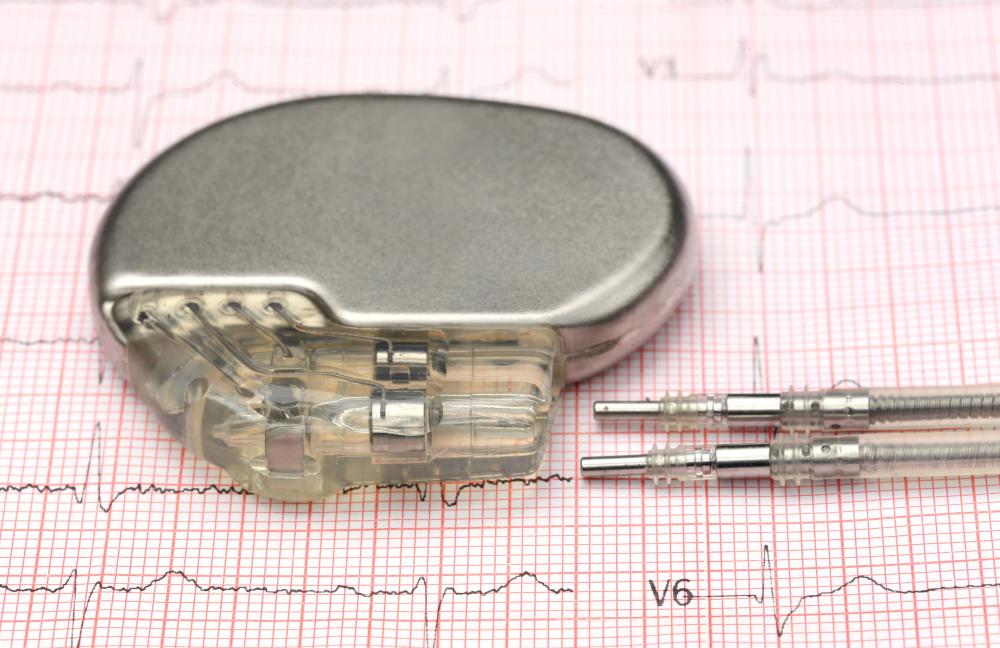
Medical Device

JH Technologies consider Medical Device Manufacturing (MDM) a sub-segment of the Microtechnology market. Catheters, stents, pacemakers, hearing aids, implantable insulin pumps, glucose meters, thermometers, biochips, and microfluidic test cells all go through the same steps of design verification, assembly, and inspection, quality assurance, and defect analysis we see in Microtechnology. MDM typically has several unique requirements driven by the Food and Drug Administration (FDA).
Although much of the equipment we provide is usable by MDM’s it also needs to be configured to meet the requirements of the customer, who is governed by the FDA. Calibration, documentation, repeatability, and reliability can be significantly more stringent in this environment.
We have worked extensively with MDM’s developing our knowledge and understanding of how to best apply our products in this market segment. Please contact us and we will be happy to provide an applications expert to help you with your specific requirement.
* If you are a customer or vendor of JH Technologies and are interested in contributing articles about a product supplied by us please contact our marketing department marketing@jhtechnologies.com or call 408-436-6336 x 302.
Related Blogs
Bruker EDS & EBSD
Recent Blogs EDS & EBSD for SEM Adding Bruker EDS to your...
Read MoreNew S lynx 2 Optical Surface Profiler
Recent Blogs New S lynx 2 Optical Surface Profiler A...
Read MoreRelated Product Categories
AFM Systems – CSI Atomic Force Microscopy
Explore high-performance x-ray inspection systems for electronics, batteries, semiconductors, and...
Read MoreSEM Detectors – EDS, EBSD, and Micro-XRF
Our portfolio covers tabletop and floor-model SEMs from Oxford Instruments...
Read MoreIndustrial X-Ray Inspection Systems | NDT Equipment
Explore high-performance x-ray inspection systems for electronics, batteries, semiconductors, and...
Read MoreSEM’s: Tabletop & Compact Floor Standing
Unlock the full potential of high-performance tabletop SEM capabilities with...
Read MoreHigh Resolution Scanning Electron Microscopes—Full Size
Unlock the full potential of high-performance tabletop SEM capabilities with...
Read MoreAccessories and Lenses
Complementing our comprehensive instrument offerings, we provide a diverse range...
Read MoreJH Analytical Services
Our analytical laboratory provides specialized testing and characterization services across...
Read MoreMicroscope Cameras And Software
In the rapidly evolving world of microscopy, having the right...
Read MoreMetrology and 3D Surface Profiling
In the realms of scientific research, industrial inspection, and quality...
Read MoreHardness Testing Solutions Testers, Blocks, And Software
We understand the importance of reliable hardness testing for quality...
Read MoreCompound Upright Microscopes
Compound microscopes for applications including education, materials manufacturing and research,...
Read MoreErgonomic Routine Stereo Microscopes
The standard in ergonomic microscopy. For users doing repeated rework...
Read MoreCompound Inverted / Metallographic Microscopes
The standard in ergonomic microscopy. For users doing repeated rework...
Read MoreMetallography Sample Preparation Equipment
We understand the complexities involved in materials preparation and analysis....
Read MoreRoutine Inspection/Assembly Microscopes
Maximize quality control and achieve flawless assembly with our durable,...
Read MoreErgonomic Research Stereo Microscopes
Reproducible results are crucial in imaging, especially when capturing intricate...
Read MoreElectron Microscopy Sample Preparation
Preparing samples flawlessly is crucial for obtaining top-notch electron microscope...
Read MoreDigital Microscope
In the era of cutting-edge technology, digital microscopes have revolutionized...
Read More